| |
Main Listing
|
|
The Carbonate Cycle
The "physical carbon pump" and the "biological carbon pump" illustrate that both the mixing of the ocean and the biological processes in the sunlit zone of the ocean are of prime importance in controlling the carbon budget of the sea and the exchange with the atmosphere. Also, we have mentioned the ways in which carbon is stored in sediments and recycled. Together, these concepts define the marine carbon cycle.
The marine carbon cycle actually involves the production and recycling of two types of carbon-rich materials: organic matter and carbonate (CaCO3). It turns out that the carbonate cycle processes about four times more carbon atoms than the organic cycle, and it is, in principle, like another pump working in reverse to the biological pump. The production of solid CaCO3 (that is, “carbonate precipitation”) occurs in the surface waters of the ocean, both organically - by organisms that build their shells from CaCO3 - and inorganically according to the chemical equilibrium in the oceans according to the following chemical equation:
Ca 2+ + 2HCO3- → CaCO3 + CO2 + H2O
It may be surprising that the deposition of large quantities of calcium carbonate from the surface ocean tends to raise the atmospheric partial pressure of CO2. However, to understand this keep in mind that carbonate precipitation is closely coupled to the "real" organic biological pump we discussed earlier. Although the linking processes are rather complex, the net effect is that the carbonate cycle acts as a kind of drag on the biological pump. The amount of drag can be modified by changing the ratio of the number of carbon atoms that are involved in the carbonate cycle to those partaking in the organic cycle.
Altering this ratio of carbon atoms can be done, for example, by changing the amount of silicate (SiO4) in seawater. If there is plenty of silicate, marine organisms called “diatoms” will grow more happily. They fix carbon into organic matter, and they take much of it down to deep waters because many diatoms, at the end of their life cycle, tend to settle out of the water where they grew. If there is very little silicate available, organisms called “coccolithophores” grow more readily than diatoms.
Coccolithophores precipitate lots of carbon into carbonate, along with making organic matter, and they, too, tend to settle out. But they remove calcium carbonate from surface waters by precipitation, which makes these waters reject carbon dioxide and thus tend to raise the atmospheric CO2 concentrations. Thus any process which tends to favor the growth of organisms made from silicate, such as diatoms, over organisms made from carbonate, such as the coccolithophorids, will tend to lower the atmospheric CO2 concentration – and vice versa - even if the total organic biomass formed in the surface layer and sinking from that layer remains constant. Factors which control the diatoms vs. coccolithophorids species include temperature, nutrient levels and light availability, but the more subtle indirect factors are not yet understood.
Let us remember at least one element concerning the carbonate cycle: Unusually intense blooms of carbonate-fixing plankton, like coccolithophores, would have the effect of bringing carbon dioxide from surface waters to the air above it – that is, increasing the atmospheric CO2 concentration. The same is true for coral and shell growth in shallow waters. We would like to know, then, what precisely causes the blooms of coccolithophores that can be seen on satellite surveys, and whether their intensity is increasing or decreasing as the planet warms. Unfortunately, this is not known at present.
Ocean Nutrient Content and Climate Change

(A) Thick Eocene radiolarians compared with (B) delicate recent ones. |
During the overall cooling of the planet, in the last 40 million years, more and more silicate has been removed from ocean in the upwelling regions around the continents (due to stronger mixing from stronger winds). We know this because radiolarians (plankton organisms using silicate to make their skeletons) have been getting thinner and more delicate through time. In the last 3 million years this process of silicate extraction has enormously accelerated, as the Antarctic Ocean started to deposit vast amounts of diatom shells. How did this alter the ratio of carbon atoms processed in the marine carbon cycle? Was there a shift toward organic matter or toward carbonate? And how would this have affected the sharing of carbon dioxide between the ocean and the atmosphere? Is this negative or positive feedback, stabilizing or destabilizing the climate? Can you begin to see the "Gaia" concept at work - how life plays a vital role in the regulation of the Earth’s atmospheric composition?
Let us now return to the organic carbon cycle (carbonate is "inorganic" because it is fixed in the minerals calcite and aragonite), and proceed to a crucial question of great interest: “How will the “productivity” of the ocean – that is, the population of phytoplankton that form the base of the marine food chain - respond to global warming?” Our simple conceptual model, balancing the addition of nutrients to the sunlit layer by upward movement with the removal of the nutrients by photosynthesis and settling contains the first hint to the answer. Two crucial factors are the vertical mixing, which moves nutrients upward, and the nutrient content of the upward-moving water, which determines how much gets into the sunlit zone. We can simply say, the productivity is determined by the rate of nutrient input into the sunlit zone, which is the product of rate of mixing and concentration of nutrients. Most of this mixing, and hence most of the nutrient input to the sunlit zone, occurs in the two-hundred mile "economic zone" next to the continents. Much of the rest of the ocean (the "blue ocean" as opposed to the "green ocean") is a desert by comparison. This does not mean that the open ocean is totally empty: just as in the deserts in San Diego County, there are plenty of things growing in the open ocean.
Vertical mixing, quite probably, is decreased when the planet warms, because mixing relies on contrasts between low and high latitudes, in temperature and precipitation. Such contrasts tend to decrease on a warm planet. Changes in nutrient content from warming are more difficult to guess. The reason is that we are talking not just about one nutrient, but about an entire array of different types of nutrients, including phosphate, nitrate, silicate, soluble iron, molybdenum, zinc and a host of other trace metals. Different species of phytoplankton need different mixtures. If any one of these is in especially short supply, it becomes "limiting" to the growth of phytoplankton.
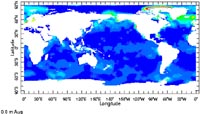
Productivity map of the surface ocean waters from satellite surveys for August. The dark blue color that represents most of the ocean area indicates low productivity (i.e. low plankton). The lighter blue color in the equatorial upwelling and coastal upwelling areas indicate higher productivity (i.e higher plankton). Greens, yellows to red color zones indicate even higher productivity (the colors in the high artic are an artifact of the measurement technique, and do not indicate high productivity in the arctic region.) |
There is little doubt that the supply of iron dust from winds will decrease with a warming of the planet (it has done so every time the climate warmed during the ice ages). Thus, in those regions where iron is limiting, productivity will decrease and so will the efficiency of the biological pump. We might also expect that the supply of silicate from the air (as minute clay particles) will decrease. In any case, a decrease in upwelling (as observed for warming) will favor calcareous plankton over siliceous plankton, increasing the "drag" on the biological pump mentioned earlier. In this aspect of the biological pump, we might expect positive feedback on warming.
|
|



